OMNICHROMA BLOCKER
These dentists recommend this product

"Amalgam tattoos and endocavities can be reliably covered with the Blocker. The layer thickness depends on the degree of discolouration and the depth of the cavity."

"My first reaction was that OMNICHROMA is truly magic. The result is also beutiful, high gloss, easy to sculpt and the procedure is simple. Except for the fact that you can skip the shade selection step your workflow is the same as with regular composites. The only addition is the blocker which you can use to cover heavy discolouration which you don´t want the composite to take it´s colour from."

"The use of the blocker requires a little bit of intuition, but even anterior restorations can be solved excellently with OMNICHROMA."
Awarded products - multiple certified service
- * Our Class IIa devices are certified as a medical device in the European Union under the Medical Device Directive 93/42/EEC by SGS CE1639, exclusively for the indication(s) detailed into the corresponding IFUs. Other non-medical uses ascribed to these devices are not within the scope of CE certification, and users should be aware product performance and/or safety has not been evaluated by SGS for those purposes.







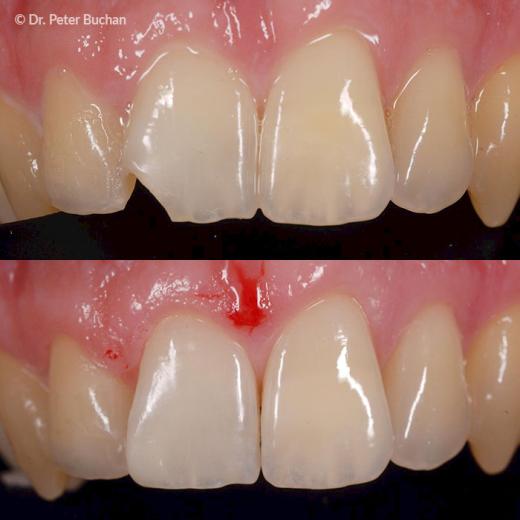
 Left: Discoloured cavity floor // Right: OMNICHROMA BLOCKER (Tokuyama Dental)
Left: Discoloured cavity floor // Right: OMNICHROMA BLOCKER (Tokuyama Dental)